A) high melting point
B) high boiling point
C) brittleness
D) poor electrical conductor when solid
E) poor electrical conductor when molten
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
In not more than three sentences, describe the electron arrangement responsible for bonding in Cl2 molecules.
Correct Answer

verified
This is covalent bonding. The chlorine a...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
In not more than three sentences, describe the key features of bonding in solid aluminum.
Correct Answer

verified
This is metallic bonding. The aluminum a...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Nitrogen and hydrogen combine to form ammonia in the Haber process. Calculate (in kJ) the standard enthalpy change H° for the reaction written below, using the bond energies given. 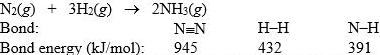
A) -969 kJ
B) -204 kJ
C) -105 kJ
D) 204 kJ
E) 595 kJ
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The lattice energy is the energy released when separated ions in the gas phase combine to form ionic molecules in the gas phase.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
No real bonds are 100% ionic in character.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on electronegativity trends in the periodic table, predict which of the following compounds will have the greatest % ionic character in its bonds.
A) H2O
B) LiI
C) CaO
D) RbF
E) HCl
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange oxygen, sulfur, calcium, rubidium, and potassium in order of decreasing electronegativity.
A) O > S > Ca > K > Rb
B) O > S > Ca > Rb > K
C) O > S > Rb > K > Ca
D) O > S > Rb > Ca > K
E) None of the above orders is correct.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the compound with the highest (i.e., most negative) lattice energy.
A) CaS(s)
B) BaO(s)
C) NaI(s)
D) LiBr(s)
E) MgO(s)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Electronegativity is a measure of
A) the energy needed to remove an electron from an atom.
B) the energy released when an electron is added to an atom.
C) the magnitude of the negative charge on an electron.
D) the attraction by an atom for electrons in a chemical bond.
E) the magnitude of the negative charge on a molecule.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is the most electronegative?
A) S
B) Ru
C) Si
D) Te
E) Cs
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Analysis of an unknown substance showed that it has a moderate melting point and is a good conductor of heat and electricity in the solid phase. Which of the following substances would have those characteristics?
A) NaCl
B) Si
C) CCl4
D) I2
E) Ga
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When two atoms form a covalently-bonded diatomic molecule, the distance between the nuclei at which the potential energy is at a minimum is called
A) the bond energy.
B) the bond length.
C) the molecular diameter.
D) the covalent radius.
E) the covalent diameter.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The lattice energy of large ions is greater in magnitude than that of small ions of the same charge.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following properties is least characteristic of typical metals?
A) moderately high melting point
B) high boiling point
C) brittleness
D) good electrical conductor when solid
E) good electrical conductor when molten
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
In covalent bond formation, the potential energy reaches a maximum when the internuclear distance is equal to the bond length.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the strongest bond in the following group.
A) C-S
B) C-O
C) C=C
D) CN
E) C-F
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following properties is least characteristic of substances composed of small, covalently-bonded molecules?
A) low melting point
B) low boiling point
C) weak bonds
D) poor electrical conductor when solid
E) poor electrical conductor when molten
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
Describe in brief how electronegativity values can be used to predict the percent ionic character of a bond between two atoms.
Correct Answer

verified
Take the electronegativity difference (F...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Describe, with appropriate explanations, the key factors which affect the magnitude of the lattice energy of an ionic substance.
Correct Answer

verified
By Coulomb's law, the energy of two elec...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 41 - 60 of 74
Related Exams