Correct Answer

verified
The bonds between carbon, C, and the hal...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the following compounds displays the greatest ionic character in its bonds?
A) NO2
B) CO2
C) H2O
D) HF
E) NH3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Acetone can be easily converted to isopropyl alcohol by addition of hydrogen to the carbon-oxygen double bond. Calculate the enthalpy of reaction using the bond energies given. 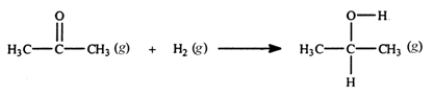

A) -484 kJ
B) -366 kJ
C) -48 kJ
D) +48 kJ
E) +366 kJ
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The lattice energy of MgCl2 is the energy change for which one of the following processes?
A) Mg(s) + Cl2(g) MgCl2(s)
B) Mg(g) + 2Cl(g) MgCl2(s)
C) Mg2+(s) + 2Cl-(g) gCl2(g)
D) Mg2+(g) + 2Cl-(g) MgCl2(s)
E) MgCl2(aq) MgCl2(s)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
A single covalent bond consists of a single delocalized electron pair.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Electronegativities on Pauling's scale are calculated from ionization energies and electron affinities.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The more C-O and O-H bonds there are in a substance, the greater will be the amount of heat released when a fixed mass of the substance is burned.
B) False
Correct Answer

verified
False
Correct Answer
verified
Multiple Choice
Quartz (SiO2) is a solid with a melting point of 1550 °C. The bonding in quartz is best described as
A) lattice energy.
B) network attractions.
C) ionic bonding.
D) covalent bonding.
E) metallic bonding.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the element whose Lewis symbol is correct.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The stronger the bonds in a fuel, the more energy it will yield.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When one mole of each of the following liquids is burned, which will produce the most heat energy?
A) C6H14
B) C5H12
C) C6H14O
D) C6H12O
E) C6H10O3
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
A hypothetical ionic substance will not form merely because it has a high lattice energy. Explain why, using energy-based arguments.
Correct Answer

verified
In order for an ionic substance to form,...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Select the compound with the lowest (i.e., least negative) lattice energy.
A) CsBr(s)
B) NaCl(s)
C) SrO(s)
D) CaO(s)
E) KBr(s)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A Born-Haber cycle applied to the formation reaction of an ionic solid
A) is normally used to calculate ionization energy.
B) is normally used to calculate electron affinity.
C) is normally used to calculate bond energy.
D) is normally used to determine the overall enthalpy change.
E) is an application of Hess's Law.
G) B) and E)
Correct Answer

verified
E
Correct Answer
verified
True/False
As a measure of the strength of metallic bonding, the boiling point of a metal is a better indicator than its melting point.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
Give a clear and concise definition of the term "electronegativity"; i.e., what does it measure?
Correct Answer

verified
Electronegativity measures an ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the following contains covalent bonds?
A) BaO
B) IBr
C) Mg
D) LiBr
E) Cu
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Essay
In not more than three sentences, describe the electron arrangement responsible for bonding in solid SrCl2.
Correct Answer

verified
This is an example of ionic bonding in which Sr, from group 2A (2) will be present as Sr2+ ions, while Cl, from group 7A (7) will be present as Cl- ions. The cations and anions will be arranged in a crystalline lattice so that nearest neighbors will be ions of opposite charge, thus achieving a net coulombic attraction. There are no molecules present; each ion is equally attracted to all its nearest neighbors.
Correct Answer
verified
Essay
Most of the copper sold in major metal markets is highly purified, typically to 99.99%. Why is this?
Correct Answer

verified
The main use of copper is as a...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Select the correct formula for a compound formed from calcium and chlorine.
A) CaCl
B) CaCl2
C) Ca2Cl
D) Ca2Cl2
E) CaCl3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 74
Related Exams