B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Atoms X, Y, Z, and R have the following nuclear compositions: 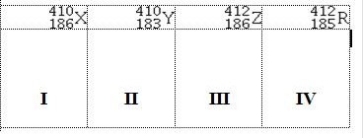 Which of the following are isotopes of the same element?
Which of the following are isotopes of the same element?
A) I & II
B) I & IV
C) II & IV
D) III & IV
E) I & III
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formula for the ionic compound formed by calcium ions and nitrate ions?
A) Ca3N2
B) Ca(NO3) 2
C) Ca2NO3
D) Ca2NO2
E) CaNO3
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Tetrasulfur dinitride decomposes explosively when heated. What is its formula?
A) S2N4
B) S4N2
C) 4SN2
D) S4N
E) S2N
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The elements in Group 7A are known by what name?
A) Transition metals
B) Halogens
C) Alkali metals
D) Alkaline earth metals
E) Noble gases
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
The empirical formula of C6H6 is CH.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The molecular formula of a molecular substance is a whole number multiple of its empirical formula.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these compounds is most likely to be ionic?
A) GaAs
B) SrBr2
C) NO2
D) CBr4
E) H2O
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a molecular formula for a compound with an empirical formula of CH?
A) C2H6
B) C3H9
C) C4H10
D) C6H6
E) None of the answers is correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formula for sodium sulfide is
A) NaS.
B) K2S.
C) NaS2.
D) Na2S.
E) SeS.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What binary compound would be formed from barium ions and fluoride ions?
A) Ba2F3
B) BaF3
C) BaF
D) Ba2F
E) BaF2
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium permanganate is a strong oxidizer that reacts explosively with easily oxidized materials. What is its formula?
A) KMnO3
B) KMnO4
C) K2MnO4
D) K(MnO4) 2
E) K2Mn2O7
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formula for the ionic compound formed by calcium and selenium?
A) CaSe
B) Ca2Se
C) CaSe2
D) Ca3Se
E) CaSe3
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Millikan's oil-drop experiment
A) established the charge on an electron.
B) showed that all oil drops carried the same charge.
C) provided support for the nuclear model of the atom.
D) suggested that some oil drops carried fractional numbers of electrons.
E) suggested the presence of a neutral particle in the atom.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a nonmetal?
A) Lithium, Li, Z = 3
B) Bromine, Br, Z = 35
C) Mercury, Hg, Z = 80
D) Bismuth, Bi, Z = 83
E) Sodium, Na, Z = 11
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the number of electrons and identify the correct symbol for an atom with 17 protons and 18 neutrons.
A) 17 electrons, ![]()
B) 18 electrons, ![]()
C) 17 electrons, ![]()
D) 17 electrons, ![]()
E) 18 electrons, ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rutherford's experiment with alpha particle scattering by gold foil established that
A) positive charge not evenly distributed throughout an atom.
B) electrons have a negative charge.
C) electrons have a positive charge.
D) atoms are made of protons, neutrons, and electrons.
E) protons are 1840 times heavier than electrons.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Who is credited with measuring the mass/charge ratio of the electron?
A) Dalton
B) Chadwick
C) Thomson
D) Millikan
E) Rutherford
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The chemical name for SO32- (aq) is sulfite ion. Therefore, the chemical name of H2SO3 (aq) is
A) dihydrosulfuric acid.
B) sulfurous acid.
C) dihydrogen sulfite.
D) hyposulfurous acid.
E) sulfuric acid.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these elements is chemically similar to oxygen?
A) Sulfur
B) Calcium
C) Iron
D) Nickel
E) Potassium
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 124
Related Exams