A) upper right
B) upper left
C) center
D) lower right
E) lower left
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species can act as a Lewis acid?
A) NH3
B) F-
C) H2O
D) NH4+
E) BF3
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
A(n) ________ can be added to a solution to increase the pH of the solution.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the concentration of OH- in a 1.0 × 10-3 M Ba(OH) 2 solution?
A) 0.50 × 10-3 M
B) 1.0 × 10-3 M
C) 2.0 × 10-3 M
D) 1.0 × 10-2 M
E) 3.3 × 10-4 M
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The following is the correct order for the acid strength for these oxoacids. HClO > HClO2 > HClO3 > HClO4
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which diagram best represents the products when equimolar amounts of HF(g) and NH3(g) react?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is an acidic oxide?
A) P4O10
B) MgO
C) Fe2O3
D) K2O
E) Cr2O3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a representation of pure liquid HX, an amphoteric substance, at equilibrium. (Each circle represents 1.0 ×10-3 mol of atoms, and the volume of the box is 1.0 L.) 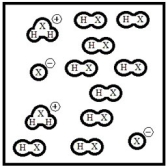 What is the autoionization constant for HX?
What is the autoionization constant for HX?
A) 4.9 ×10-2
B) 4.0 ×10-3
C) 4.4 ×10-4
D) 4.0 ×10-6
E) 2.5 ×105
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the formula for the hydronium ion?
A) OH-
B) H2O
C) H3O+
D) H3O-
E) H2O+
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a representation of an aqueous solution of a weak acid HA at equilibrium. (Each circle represents 1.0 ×10-3 mol of atoms, and the volume of the box is 1.0 L. Solvent water molecules are not shown for clarity.) 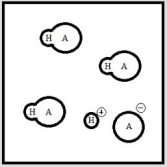 What is the pH of the solution?
What is the pH of the solution?
A) 2.4
B) 3.0
C) 2.1
D) 11.0
E) 11.6
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
All strong acids have weak conjugate bases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which aqueous solution has the highest pH?
A) 0.1 M CH3COOH, pKa = 4.7
B) 0.1 M CuCl2, pKa = 7.5
C) 0.1 M H3C6H5O7, pKa = 3.1
D) 0.1 M ZnCl2, pKa = 9.0
E) pure water
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Farmers who raise cotton once used arsenic acid, H3AsO4, as a defoliant at harvest time. Arsenic acid is a polyprotic acid with Ka1 = 2.5 × 10-4, Ka2 = 5.6 × 10-8, and Ka3 = 3 × 10-13. What is the pH of a 0.500 M solution of arsenic acid?
A) 0.85
B) 1.95
C) 3.90
D) 4.51
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
HCN is classified as a weak acid in water. What does this classification mean?
Correct Answer

verified
A relatively small f...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
What is the conjugate acid of NH3?
A) NH2
B) NH2+
C) NH2-
D) NH4
E) NH4+
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Acid strength decreases in the series HI > HSO4-> HF > HCN. Which of these anions is the weakest base?
A) I-
B) SO42-
C) F-
D) CN-
F) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
A hydrohalic acid is a binary acid containing a halogen.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Amphoteric oxides exhibit both acidic and basic properties.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a 0.014 M Ca(OH) 2 solution?
A) 1.85
B) 1.55
C) 12.15
D) 12.45
E) 15.85
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name given to a substance that can act as a Brønsted acid or as a Brønsted base according to what it is reacting with?
A) hydrophilic
B) hydrophobic
C) amphoteric
D) isoprotic
E) isoelectronic
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 137
Related Exams