A) Alkali metals
B) Noble gases
C) Halogens
D) Alkaline earth metals
E) Metalloids
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is ionic?
A) PF 3
B) CS 2
C) HCl
D) SO 2
E) MgCl 2
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a Millikan oil-drop experiment, the charges on several different oil drops were as follows: −5.92; −4.44; −2.96; −8.88. The units are arbitrary. What is the likely value of the electronic charge in these arbitrary units?
A) −1.11
B) −1.48
C) −2.22
D) −2.96
E) −5.55
G) None of the above
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
J. J. Thomson studied cathode ray particles (electrons) and was able to measure the mass/charge ratio. His results showed that
A) the mass/charge ratio varied with as the cathode material was changed.
B) the charge was always a whole-number multiple of some minimum charge.
C) matter included particles much smaller than the atom.
D) atoms contained dense areas of positive charge.
E) atoms are largely empty space.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of BBr3?
A) Boron bromide
B) Boric bromide
C) Boron tribromide
D) Tribromoboride
E) Bromine triboride
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which, if any, of the following elements do not occur in the major classes of organic compounds?
A) H
B) C
C) N
D) O
E) All of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The substance, CoCl2, is useful as a humidity indicator because it changes from pale blue to pink as it gains water from moist air. What is its name?
A) Cobalt dichloride
B) Cobalt(II) chloride
C) Cobalt chloride
D) Cobaltic chloride
E) Copper(II) chloride
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium chromate is used to protect iron from corrosion and rusting. Determine its molecular mass.
A) 261.97 amu
B) 238.98 amu
C) 161.97 amu
D) 138.98 amu
E) 74.99 amu
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Compound 1 has a composition of 46.7 mass % of element A and 53.3 mass % of element B. A and B also form a second binary compound (compound 2) . If the compositions of the two compounds are consistent with the law of multiple proportions, which of the following compositions could be that of compound 2?
A) 23.4 mass % A 76.6 mass % B
B) 30.4 mass % A 69.6 mass % B
C) 33.3 mass % A 66.7 mass % B
D) 53.3 mass % A 46.7 mass % B
E) 73.3 mass % A 26.7 mass % B
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following combinations of names and formulas is incorrect?
A) H 3PO 4 phosphoric acid
B) HNO 3 nitric acid
C) NaHCO 3 sodium carbonate
D) H 2CO 3 carbonic acid
E) KOH potassium hydroxide
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rutherford bombarded gold foil with alpha (α) particles and found that a small percentage of the particles were deflected. Which of the following was not accounted for by the model he proposed for the structure of atoms?
A) The small size of the nucleus
B) The charge on the nucleus
C) The total mass of the atom
D) The existence of protons
E) The presence of electrons outside the nucleus
G) A) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Bromine is the only nonmetal that is a liquid at room temperature. Consider the isotope bromine-81, 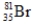 . Select the combination which lists the correct atomic number, neutron number, and mass number, respectively.
. Select the combination which lists the correct atomic number, neutron number, and mass number, respectively.
A) 35, 46, 81
B) 35, 81, 46
C) 81, 46, 35
D) 46, 81, 35
E) 35, 81, 116
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of the acid formed when HCN gas is dissolved in water?
A) Cyanic acid
B) Hydrocyanic acid
C) Cyanous acid
D) Hydrocyanous acid
E) Hydrogen cyanide
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The compound, (NH4) 2S, can be used in analysis for trace amounts of metals present in a sample. What is its name?
A) Ammonium sulfide
B) Diammonium sulfide
C) Ammonium sulfite
D) Ammonia(I) sulfite
E) Ammonium(I) sulfide
G) All of the above
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which of the following compounds is covalent?
A) CaCl 2
B) MgO
C) Al 2O 3
D) Cs 2S
E) PCl 3
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following symbols does not represent an element?
A) O 2
B) Co
C) HF
D) Cs
E) Xe
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a nonmetal?
A) Lithium, Li, Z = 3
B) Bromine, Br, Z = 35
C) Mercury, Hg, Z = 80
D) Bismuth, Bi, Z = 83
E) Sodium, Na, Z = 11
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The colorless substance, MgF2, is used in the ceramics and glass industry. What is its name?
A) Magnesium difluoride
B) Magnesium fluoride
C) Magnesium(II) fluoride
D) Monomagnesium difluoride
E) None of these choices are correct
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a metalloid?
A) Carbon, C, Z = 6
B) Sulfur, S, Z = 16
C) Germanium, Ge, Z = 32
D) Iridium, Z = 77
E) Bromine, Br, Z = 35
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Modern studies have shown that the Law of Multiple Proportions is not valid.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 101
Related Exams