A) same number of protons
B) same number of protons and electrons
C) same number of neutrons
D) same number of neutrons and electrons
F) A) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by humans and other vertebrates, but not by other organisms such as bacteria or plants?
A) calcium
B) iodine
C) sodium
D) phosphorus
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What coefficients must be placed in the following blanks so that all atoms are accounted for in the products? C₆H₁₂O₆ → ________ C₂H₆O + ________ CO₂
A) 2; 1
B) 3; 1
C) 1; 3
D) 2; 2
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
a(n. ________ has charge but negligible mass, whereas a(n) ________ has mass but no charge.
A) proton; neutron
B) neutron; proton
C) neutron; electron
D) electron; neutron
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When are atoms most stable?
A) when they have the fewest possible valence electrons
B) when they have the maximum number of unpaired electrons
C) when all of the electron orbitals in the valence shell are filled
D) when all electrons are paired
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following structure where A and B represent two different elements, the valency of A is ________ and B is ________.
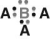
A) one; three
B) one; five
C) three; five
D) eight; eight
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
About 25 of the 92 natural elements are known to be essential to life. Which 4 of these 25 elements make up approximately 96% of living matter?
A) carbon, sodium, hydrogen, nitrogen
B) carbon, oxygen, phosphorus, hydrogen
C) oxygen, hydrogen, calcium, nitrogen
D) carbon, hydrogen, nitrogen, oxygen
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An ion with six protons, seven neutrons, and a charge of 2+ has an atomic number of ________.
A) four
B) five
C) six
D) seven
F) A) and B)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Compared with ³¹P, the radioactive isotope ³²P has
A) a different atomic number.
B) one more proton.
C) one more electron.
D) one more neutron.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Can the atomic mass of an element vary?
A) No, it is fixed; otherwise a new element will be formed.
B) Yes. Adding or losing electrons will substantially change the atomic mass.
C) Yes. Adding or losing protons will change the atomic mass without forming a different element.
D) Yes. Adding or losing neutrons will change the atomic mass without forming a different element.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Molybdenum has an atomic number of 42. Several common isotopes exist, with mass numbers from 92-100. Which of the following can be true?
A) Molybdenum atoms can have between 50 and 58 neutrons.
B) Molybdenum atoms can have between 50 and 58 protons.
C) Molybdenum atoms can have between 50 and 58 electrons.
D) Isotopes of molybdenum have different numbers of electrons.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following are compounds?
A) H₂O, O₂, and CH₄
B) H₂O and O₂
C) O₂ and CH₄
D) H₂O and CH₄, but not O₂
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Carbon-14 has the same ________.
A) atomic number and atomic mass as carbon-12
B) atomic number and thus number of neutrons as carbon-13
C) atomic mass as both carbon-12 and carbon-13
D) number of protons but more neutrons than carbon-12
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Refer to the following figure to answer the questions below.
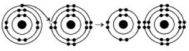 -When the atoms involved in a covalent bond have the same electronegativity, what type of bond results?
-When the atoms involved in a covalent bond have the same electronegativity, what type of bond results?
A) an ionic bond
B) a hydrogen bond
C) a nonpolar covalent bond
D) a polar covalent bond
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Refer to the following figure to answer the questions below.
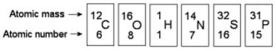 -How many electrons will a single atom of sulfur with no charge and no bonds have in its valence shell?
-How many electrons will a single atom of sulfur with no charge and no bonds have in its valence shell?
A) 6
B) 8
C) 16
D) 32
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Refer to the following figure to answer the questions below.
3H₂ + N₂ 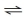 2NH₃
-Which of the following is true for the reaction?
2NH₃
-Which of the following is true for the reaction?
A) The reaction is nonreversible.
B) Hydrogen and nitrogen are the reactants of the reverse reaction.
C) Ammonia is being formed and decomposed simultaneously.
D) Only the forward or reverse reactions can occur at one time.
F) None of the above
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Refer to the following figure to answer the questions below.
 -Oxygen has an atomic number of 8 and, most commonly, a mass number of 16. Thus, what is the atomic mass of an oxygen atom?
-Oxygen has an atomic number of 8 and, most commonly, a mass number of 16. Thus, what is the atomic mass of an oxygen atom?
A) approximately 8 grams
B) approximately 8 daltons
C) approximately 16 grams
D) approximately 16 daltons
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Refer to the following figure to answer the questions below.
 -What is the atomic number of the cation formed in the reaction in the illustration?
-What is the atomic number of the cation formed in the reaction in the illustration?
A) 8
B) 10
C) 11
D) 16
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Atoms have no electric charge because they have ________.
A) uncharged neutrons in their nuclei
B) an equal number of protons and neutrons
C) an equal number of protons and electrons
D) an equal number of charged and uncharged subatomic particles
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which bond or interaction would be difficult to disrupt when compounds are put into water?
A) covalent bonds between carbon atoms
B) hydrogen bonds
C) ionic bonds
D) ionic and hydrogen bonds
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 61
Related Exams