B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the expression below, showing the terms which contribute to the heat of solution, ΔHsoln: ΔHsoln = ΔHsolute + ΔHsolvent + ΔHmix Which of the following sets correctly shows the signs (positive or negative) of the three terms on the right hand side of the equation?
A) ΔHsolute > 0; ΔHsolvent > 0; ΔHmix > 0
B) ΔHsolute < 0; ΔHsolvent < 0; ΔHmix < 0
C) ΔHsolute > 0; ΔHsolvent > 0; ΔHmix < 0
D) ΔHsolute < 0; ΔHsolvent < 0; ΔHmix > 0
E) ΔHsolute < 0; ΔHsolvent > 0; ΔHmix < 0
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The concentration of iodine in sea water is 60. parts per billion by mass. If one assumes that the iodine exists in the form of iodide anions, what is the molarity of iodide in sea water? (The density of sea water is 1.025 g/mL.)
A) 4.8 × 10-13 M
B) 4.8 × 10-10 M
C) 4.8 × 10-7 M
D) 4.7 × 10-4 M
E) 4.7 × 10-1 M
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Methane has a Henry's Law constant (k) of 9.88 × 10-2 mol/(L·atm) when dissolved in benzene at 25°C. How many grams of CH4 will dissolve in 3.00 L of benzene if the partial pressure of CH4 is 1.48 atm?
A) 0.0667 g
B) 0.146 g
C) 2.34 g
D) 4.83 g
E) 7.02 g
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An emulsion is a dispersion consisting of a
A) solid in a liquid.
B) liquid in a liquid.
C) gas in a liquid.
D) liquid in a solid.
E) gas in a solid.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Safrole is used as a topical antiseptic. Calculate the vapor pressure of a solution prepared by dissolving 0.75 mol of safrole in 950 g of ethanol ( 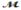 = 46.07 g/mol) . P°ethanol = 50.0 torr at 25°C.
= 46.07 g/mol) . P°ethanol = 50.0 torr at 25°C.
A) 1.8 torr
B) 11 torr
C) 15 torr
D) 40 torr
E) 48 torr
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume of concentrated (14.7 M) phosphoric acid is needed to prepare 25.0 L of 3.0 M H3PO4?
A) 0.20 L
B) 0.57 L
C) 1.8 L
D) 3.6 L
E) 5.1 L
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aqueous ammonia is commercially available in a solution that is 28% (w/w) ammonia. What is the mole fraction of ammonia in such a solution?
A) 0.017
B) 0.023
C) 0.012
D) 0.24
E) 0.29
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the molarity of a solution prepared by diluting 1.85 L of 6.5 M KOH to 11.0 L.
A) 0.28 M
B) 0.91 M
C) 1.1 M
D) 3.1 M
E) 3.9 M
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Copper(II) bromide is used as a wood preservative. What mass of CuBr2 is needed to prepare 750.0 mL of a 1.25 M solution?
A) 134 g
B) 209 g
C) 372 g
D) 938 g
E) > 1 kg
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Two aqueous solutions are prepared: 2.0 m Cu(NO3) 2 and 2.0 m NaBr. Which of the following statements is true?
A) The Cu(NO3) 2 solution has a higher vapor pressure and lower freezing point than the NaBr solution.
B) The Cu(NO3) 2 solution has a higher vapor pressure and higher freezing point than the NaBr solution.
C) The Cu(NO3) 2 solution has a lower vapor pressure and lower freezing point than the NaBr solution.
D) The Cu(NO3) 2 solution has a lower vapor pressure and higher freezing point than the NaBr solution.
E) None of these choices are correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When two pure substances are mixed to form a solution
A) heat is released.
B) heat is absorbed.
C) there is an increase in entropy.
D) there is a decrease in entropy.
E) entropy is conserved.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Saccharin, one of the first non-nutritive sweeteners used in soft-drinks, is 500 times sweeter than sugar in dilute aqueous solutions. The solubility of saccharin is 1.00 gram per 290 mL of solution. What is the molarity of a saturated saccharin solution? ( 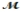 saccharin = 183.2 g/mol)
saccharin = 183.2 g/mol)
A) 0.0188 M
B) 0.632 M
C) 1.58 M
D) 3.45 M
E) None of these choices are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cinnamaldehyde ( 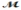 = 132.15 g/mol) is used as a flavoring agent. What mass of cinnamaldehyde must be added to 175 g of ethanol to give a solution whose boiling point is 82.7°C? Kb = 1.22°C/m, boiling point of pure ethanol = 78.5°C
= 132.15 g/mol) is used as a flavoring agent. What mass of cinnamaldehyde must be added to 175 g of ethanol to give a solution whose boiling point is 82.7°C? Kb = 1.22°C/m, boiling point of pure ethanol = 78.5°C
A) 62.4 g
B) 67.8 g
C) 76.2 g
D) 78.5 g
E) 79.6 g
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The chemist, Anna Lytic, must prepare 1.00 kg of 15.0% (w/w) acetic acid using a stock solution which is 36.0% (w/w) acetic acid (d = 1.045 g/mL) . Which of the following combinations will give her the solution she wants?
A) 417 mL of 36% acetic acid in 583 mL of distilled water
B) 417 g of 36% acetic acid in 583 g of distilled water
C) 360 mL of 36% acetic acid in 640 mL of distilled water
D) 360 g of 36% acetic acid in 640 g of distilled water
E) 150 g of 36% acetic acid in 850 g of distilled water
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following aqueous solutions should demonstrate the most non-ideal behavior?
A) 0.1 M NaI
B) 0.2 M CuSO4
C) 0.5 M KBr
D) 2.0 M CuCl2
E) 2.0 M CsCl
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
In general, water is a good solvent for both polar and nonpolar compounds.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mole fraction of potassium nitrate in an aqueous solution is 0.0194. The solution's density is 1.0627 g/mL. Calculate the molarity of the solution.
A) 0.0194 M
B) 0.981 M
C) 1.05 M
D) 1.96 M
E) 19.4 M
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.100 m K2SO4 solution has a freezing point of -0.43°C. What is the van't Hoff factor for this solution? Kf = 1.86°C/m
A) 0.77
B) 1.0
C) 2.3
D) 3.0
E) >3.0
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Raoult's Law relates the vapor pressure of the solvent above the solution to its mole fraction in the solution. Which of the following is an accurate statement?
A) Raoult's Law applies exactly to all solutions.
B) Raoult's Law works best when applied to concentrated solutions.
C) Raoult's Law works best when applied to dilute solutions.
D) Raoult's Law applies only to non-ideal solutions.
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 96
Related Exams