A) a 1,3-diketopentanoate.
B) a diethyl malonate.
C) an ethyl acetoacetate.
D) a b-keto ester.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the starting material for the following reaction? 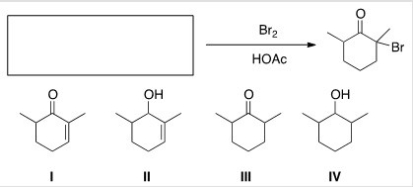
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following four compounds is the most acidic? 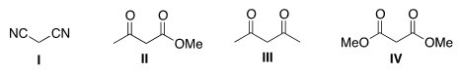
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the most acidic proton in the following compound? 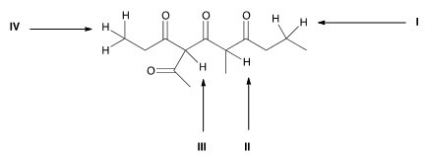
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why can't you prepare 2-tert-butylcyclohexanone by the following reaction? 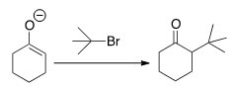
A) Because tert-butyl bromide is too basic.
B) Because tert-butyl bromide cannot undergo an SN2 reaction.
C) Because tert-butyl bromide is a nucleophile.
D) Because tert-butyl bromide is not a stable compound.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For most compounds with a single keto group in the molecule,equilibrium favors the keto form over the enol form of the compound.This is due largely to what?
A) The C=O bond is much stronger than the C=C bond.
B) The C=C bond is much stronger than the C=O bond.
C) The keto form can undergo intramolecular hydrogen bonding.
D) The enol form can undergo intramolecular hydrogen bonding.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the three steps in the malonic ester synthesis?
A) Deprotonation,alkylation,hydrolysis/decarboxylation
B) Hydrogenation,alkylation,deprotonation
C) Alkylation,hydrolysis/decarboxylation.hydrogenation
D) Hydrolysis/decarboxylation,deprotonation,alkylation
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing reagent in the reaction below? 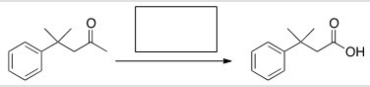
A) Br2/AcOH
B) I2/KOH
C) I2
D) KOH
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the least acidic compound? 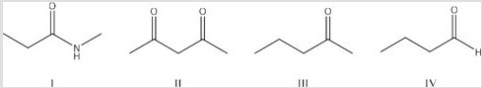
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an enol form of the following compound? 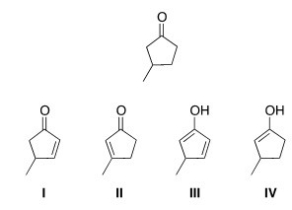
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is the most acidic?
A) Ethyl acetoacetate
B) 2-Butanone
C) 1-Butanol
D) 3-Pentanone
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds would undergo racemization in the presence of a base? 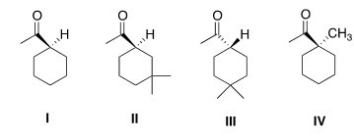
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the enolate of acetone less basic than the allyl anion derived from propene?
A) Because there are more atoms in acetone
B) Because there are more resonance structures for the enolate of acetone
C) It isn't; the allyl anion is less basic.
D) One of the resonance structures for the enolate places the negative charge on the more electronegative oxygen.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Starting with cyclohexanone,how could you prepare the diketone below? 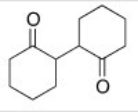
A) Treat cyclohexanone with a base under thermodynamic conditions.
B) Hydrogenate cyclohexanone with Raney nickel.
C) Convert cyclohexanone into the a-bromoketone and then react this with the enolate of cyclohexanone.
D) Convert cyclohexanone into an enamine with diethylamine and then react this with more cyclohexanone.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
It has been found that b-dicarbonyl compounds have a greater concentration of the enol form over the keto form.This can be explained by
A) the C=C of the enol is conjugated with the carbonyl group.
B) the -OH of the enol can hydrogen bond to the oxygen of the nearby carbonyl group.
C) Both and above are true.
D) None of the choices are true.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Treatment of 2-hexanone with NaOCH2CH3 followed by CH3Br affords compound X (C7H14O) as the major product.X shows a strong absorption in the IR spectrum at 1713 cm-1,and its 1H NMR data is given below.What is the structure of X? 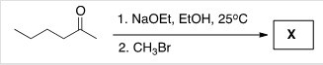


A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If you want to form a kinetic enolate,you want to
A) use a strong,non-nucleophilic base such as LDA.
B) use a protic solvent.
C) use a low temperature.
D) both use a strong,non-nucleophilic base such as LDA and use a low temperature.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following bases will completely convert 1,4-cyclohexandione into an enolate?
A) Sodium hydroxide
B) Sodium methoxide
C) Sodium tert-butoxide
D) Sodium hydride
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing reagent for the following reaction? 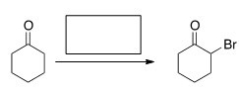
A) Br2/HOAc
B) Br2/KOH
C) Cl2/FeCl3
D) Br2/FeBr3
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Will acetophenone be completely deprotonated by lithium diisopropylamide (LDA) ?
A) Yes
B) No
D) undefined
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 47
Related Exams